Unveiling the Versatility of Vesicular Stomatitis Virus: Pseudotyping and Beyond
Vesicular stomatitis virus (VSV), a bullet-shaped, enveloped virus belonging to the Rhabdoviridae family, possesses a distinctive single-stranded, negative-sense RNA genome encoding five vital proteins: nucleoprotein (N), phosphoprotein (P), matrix protein (M), glycoprotein (G), and large polymerase protein (L). With the G protein situated on the viral envelope, VSV naturally infects diverse mammals, from cattle and horses to pigs and rodents.
VSV's simplicity and well-explored biology render it an invaluable model for delving into fundamental virological processes. Researchers have extensively utilized VSV to unravel viral replication, transcription, and translation as well as host-pathogen interactions, catalyzing breakthroughs in cellular and molecular biology.
Unraveling the Concept of Pseudotyping
One standout application of VSV lies in pseudotyping, where the native G protein is swapped with an envelope protein from another virus. Pseudotyping hinges on a seemingly simple yet transformative process. By substituting the envelope protein of one virus with that of another, pseudotyped viral particles are born—bearing the genetic material of one virus and the surface characteristics of another. This envelope protein governs the virus's capacity to adhere to and infiltrate specific host cells that may normally remain beyond the virus’s reach. Pseudotyping also offers a safer means to scrutinize viral entry mechanisms, especially when tackling highly pathogenic viruses, including HIV, SARS-CoV-2, and influenza. VectorBuilder was able to take advantage of this opportunity to be the first company to offer SARS-CoV-2 pseudotyped virus during the COVID-19 pandemic. This controlled approach enables researchers to dissect viral behavior and interactions, paving the way for diagnostic tools, therapies, and vaccine development.
When applied to VSV, pseudotyping uncovers a realm of possibilities that contribute significantly to the field of virology and biomedical innovation. VSV's innate ability to adapt, replicate robustly, and easily accommodate foreign envelope proteins bestows it with unique advantages. Pseudotyped viral particles retain VSV's effective replication machinery while adopting the host cell tropism and entry characteristics of the chosen envelope protein. Beyond insight into viral biology, VSV pseudotyped particles find application in gene delivery, vaccine development, and antiviral drug screening, showcasing the versatility of pseudotyping VSV to advance our understanding of virology and fuel innovative therapeutic strategies.

Figure 1. Cartoon diagram of Wild-type VSV, VSV without surface G protein, and VSV psuedotyped with the spike protein of another virus.
In a study published in 2020, researchers focused on the utilization of pseudotyped viruses, specifically vesicular stomatitis virus (VSV), as an alternative approach to investigate neutralizing antibodies against dangerous viruses like Ebola virus (EBOV). The study utilized EBOV G protein inserted onto both HIV-1 and VSV platforms to provide insights into neutralization responses. The findings revealed the potential of pseudotyped virus assays in evaluating vaccine responses and therapeutic efficacy. While both HIV-1 and VSV-based assays showed positive correlations with live EBOV neutralization, the VSV-based assay demonstrated better sensitivity, specificity, and correlation. The study underscores the importance of optimizing pseudotyped virus neutralization assays, considering specific viral envelope proteins, to enhance their reliability in assessing vaccine effectiveness and therapeutic interventions against dangerous viruses like Ebola.
Vesicular Stomatitis Virus Beyond Pseudotyping
Beyond its pivotal role in pseudotyping, VSV has demonstrated remarkable versatility and potential in diverse areas of virology and biomedical research. One of the most promising applications is in oncolytic virotherapy, where engineered VSV strains are harnessed to target and selectively destroy cancer cells. The virus's innate ability to preferentially replicate in tumor cells, triggering their lysis while sparing healthy tissue, has led to ongoing investigations into VSV-based cancer treatments. Additionally, VSV's capacity to induce immune responses and stimulate anti-tumor immunity further amplifies its attractiveness in the realm of cancer therapeutics.
A recent phase 1 clinical trial has brought to the forefront the potential of VSV as a promising treatment for relapsed refractory lymphoma. This innovative trial centered on the administration of a sole intravenous dose of VSV-IFNβ-NIS, recombinant VSV expressing interferon-β (IFN-β) and sodium iodide symporter (NIS), to patients across a spectrum of dose levels. Encouragingly, the outcomes not only demonstrated safety and efficacy without encountering dose-limiting toxicities but also showcased pronounced success, particularly in patients afflicted by T-cell lymphoma (TCL). Remarkably, a subset of TCL patients exhibited partial or complete remissions.
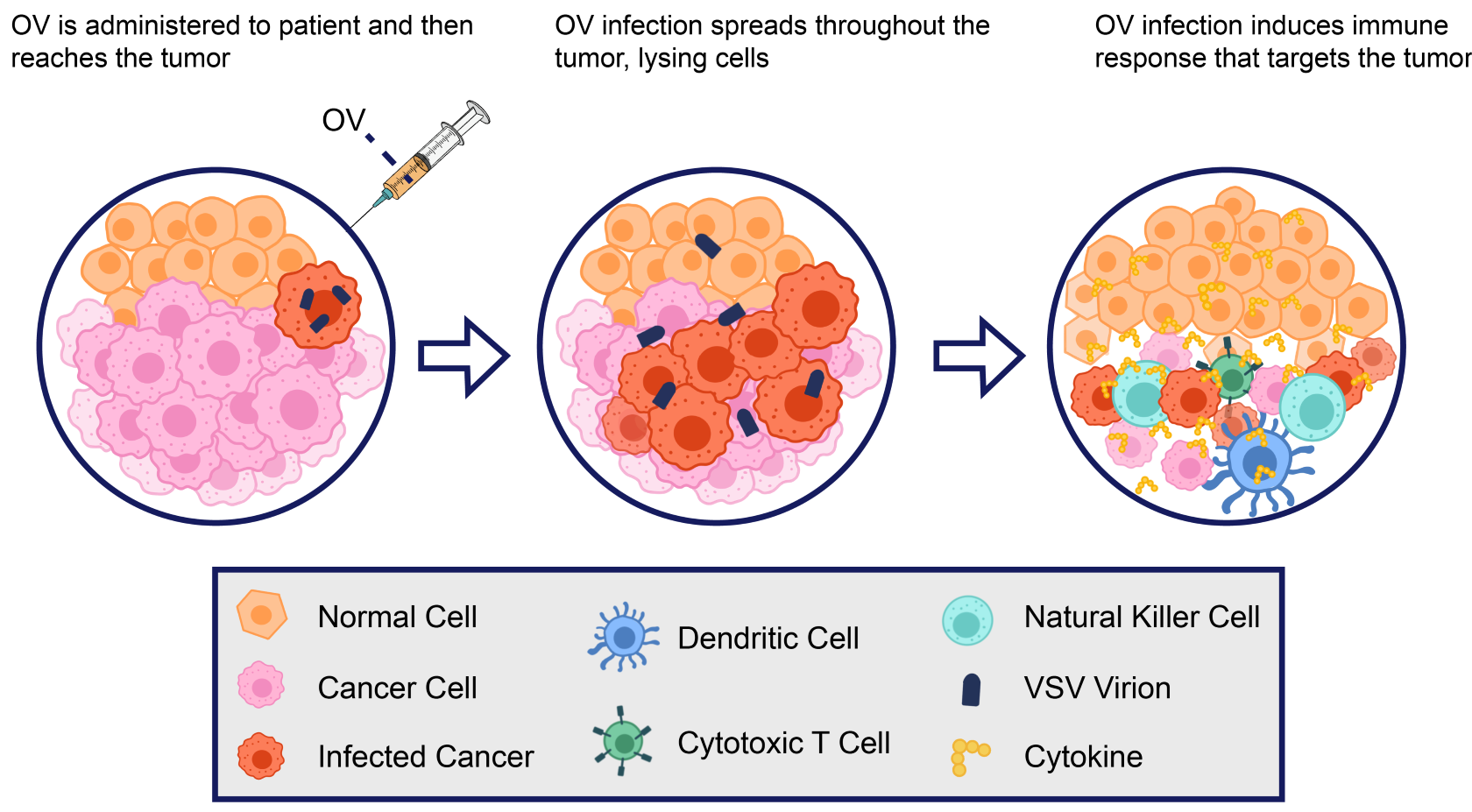
Figure 2. Mechanism of action of VSV oncolytic therapy.
In addition to oncolytic therapy, VSV's neurotropic properties have opened avenues for significant advancements in neuroscientific research. The virus's ability to infect neurons and trace neural pathways has facilitated intricate mapping of brain circuits, enabling researchers to unravel complex neural connections. In the study of neurological disorders, VSV-based approaches have been utilized to model and investigate conditions like Alzheimer's, Parkinson's, and prion diseases. By delivering therapeutic genes or modulating neural activity, VSV offers a unique toolset for exploring neurobiology and developing potential interventions for neurological ailments.
These multifaceted applications of VSV highlight its capacity to transcend the boundaries of pseudotyping and study of viral entry, making lasting contributions to fields ranging from oncology to neuroscience, underscoring its significance as a versatile and powerful research tool. This system is also continuing to prove its sustainability as a platform for combatting and preventing disease. As the boundaries of VSV's capabilities continue to expand, its potential is only bound by researchers’ imaginations.
Sources
Steeds, K., Hall, Y., Slack, G.S. et al. Pseudotyping of VSV with Ebola virus glycoprotein is superior to HIV-1 for the assessment of neutralising antibodies. Sci Rep 10, 14289 (2020). https://doi.org/10.1038/s41598-020-71225-1
Cook J, Peng KW, Witzig TE, Broski SM, Villasboas JC, Paludo J, Patnaik M, Rajkumar V, Dispenzieri A, Leung N, Buadi F, Bennani N, Ansell SM, Zhang L, Packiriswamy N, Balakrishnan B, Brunton B, Giers M, Ginos B, Dueck AC, Geyer S, Gertz MA, Warsame R, Go RS, Hayman SR, Dingli D, Kumar S, Bergsagel L, Munoz JL, Gonsalves W, Kourelis T, Muchtar E, Kapoor P, Kyle RA, Lin Y, Siddiqui M, Fonder A, Hobbs M, Hwa L, Naik S, Russell SJ, Lacy MQ. Clinical activity of single-dose systemic oncolytic VSV virotherapy in patients with relapsed refractory T-cell lymphoma. Blood Adv. 2022 Jun 14;6(11):3268-3279. doi: 10.1182/bloodadvances.2021006631. PMID: 35175355; PMCID: PMC9198941.



